To Mass Produce Propane C3h8 a Continuously Stirred Tank Reactor Cstr Is Used
Question
Reaction Engineering
Propylene (C3H6) is a valuable feed stock which is commonly produced in gas phase by thermal cracking of Propane (C3H8). Hydrogen in gas phase is also formed in this process. The reaction is carried out in an ideal reactor as shown below:
\mathrm{C}_{3} \mathrm{H}_{8} \rightarrow \mathrm{C}_{3} \mathrm{H}_{6}+\mathrm{H}_{2}
The reaction rate equation is of the first order with respect to Propane (A): -rA=k.CA
It is required to produce Propylene with 80% conversion when the volumetric flow rate of Propane is 50 L/min, the initial concentration of A (CAO) is 10 mol/L, and the rate constant (k) is 0.5 min-1. Assume that reaction occurs under isothermal conditions.
O Calculate the fractional volume change of A (EA), the final concentration of A(CA) and final molar flow rate (mol/min) of A (FA) at the exit of the reactor.
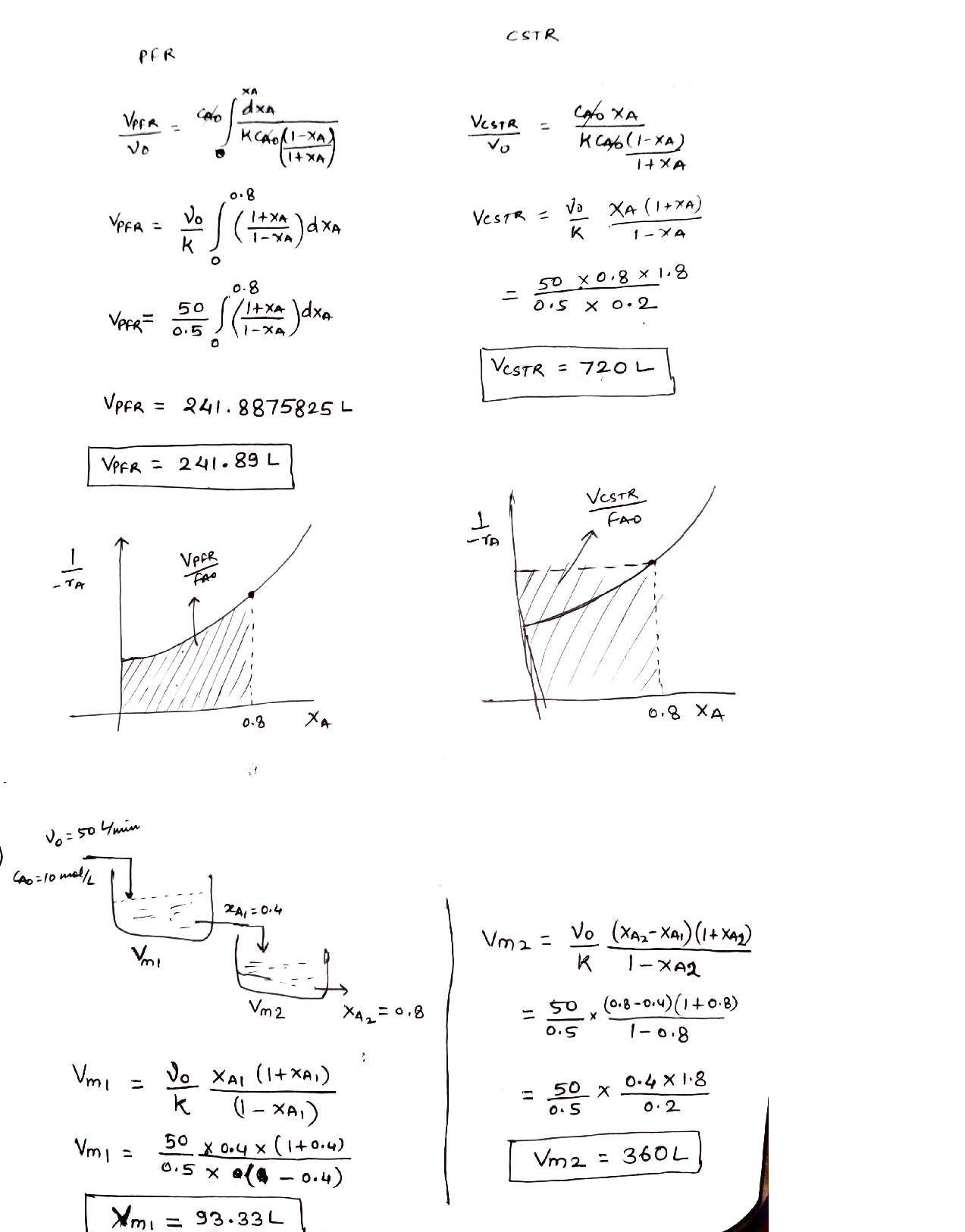
Provide a comparison and evaluation of the volumes of a PFR reactor (VPFR)and a CSTR reactor (VCSTR) for the reaction.
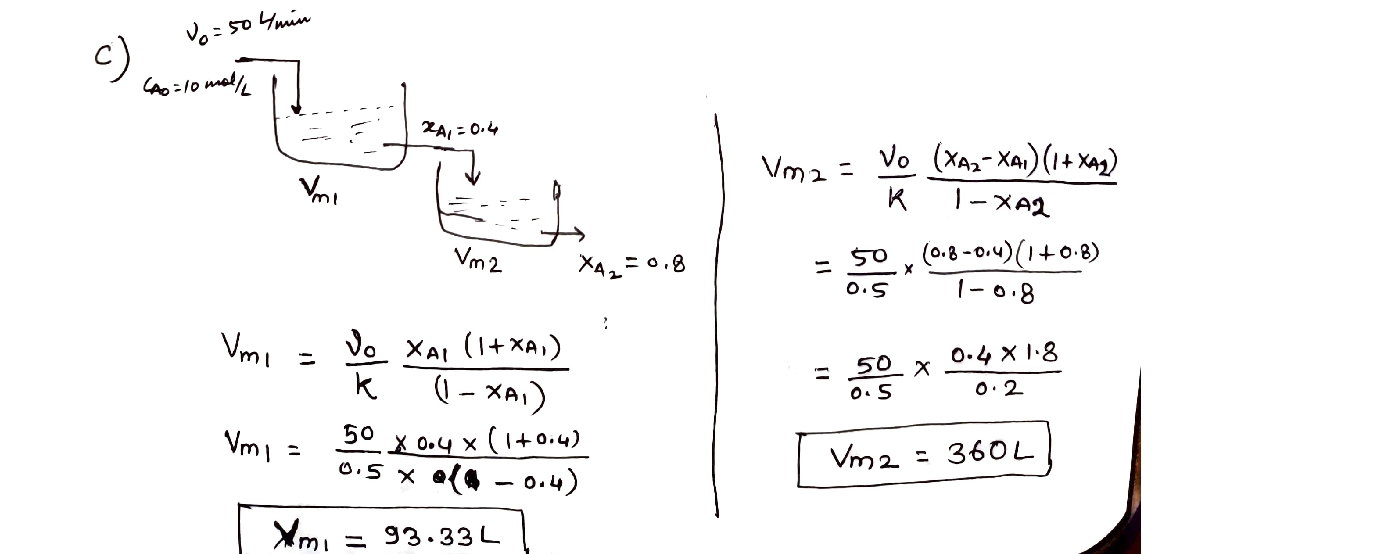
If the reaction is carried out in two CSTR in series with 40% conversion in the first reactor and the overall conversion of 80% of the reactant (A),estimate the volumes of two CSTR.[10%]
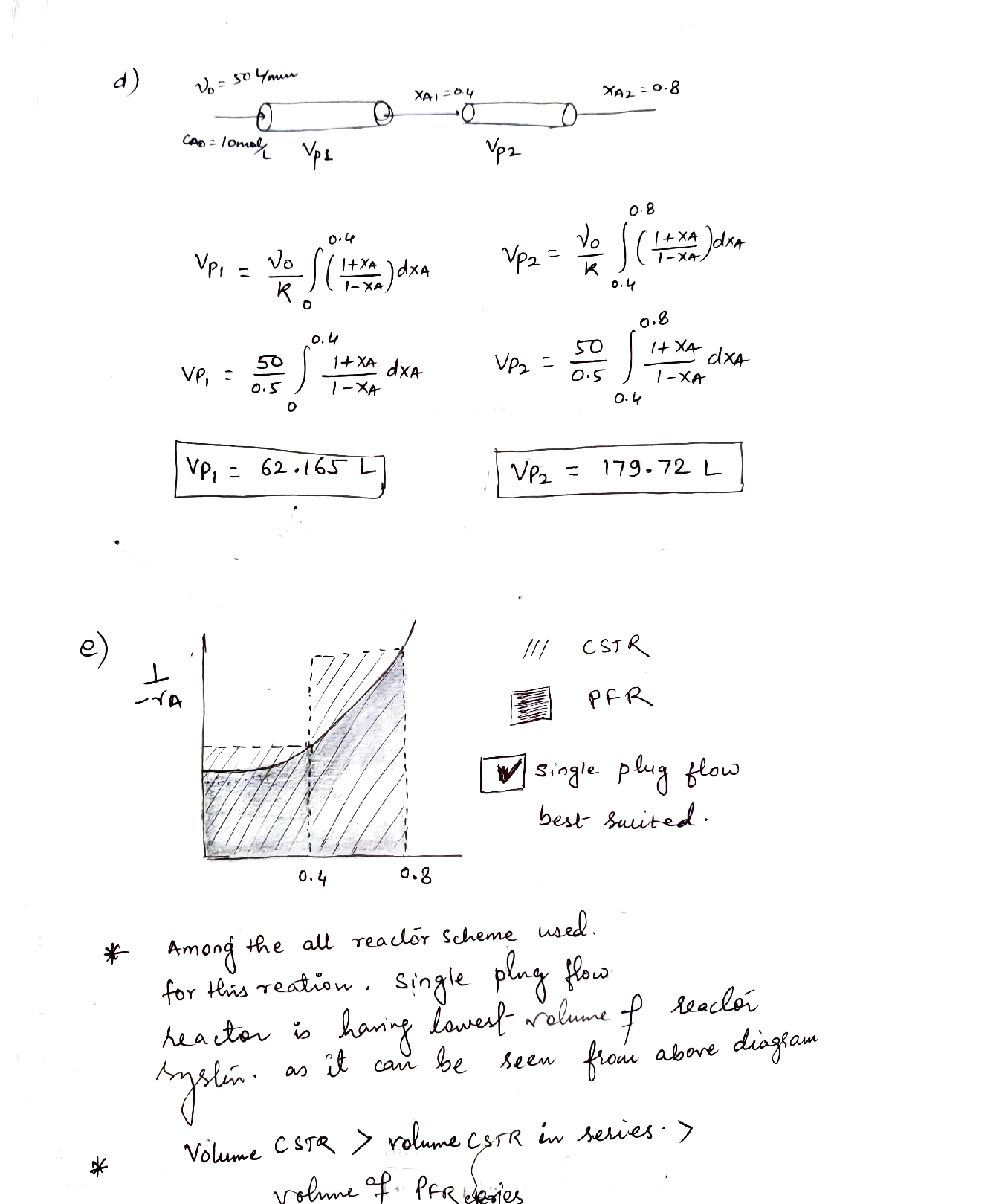
If the reaction is carried out in two PFR in series under the same conditions as the above item c, estimate the volumes of two PFR.[10%]
) Suggest other suitable alternatives (if possible) and the best type of reactor system for the given reaction, to achieve the desired conversion of 0.8. Give Two reasons to support your explanation. You may support your solution(s) by the aid of diagrams and calculations.[10%]
Answer
Verified

Submit query
Getting answers to your urgent problems is simple. Submit your query in the given box and get answers Instantly.
Success
Source: https://tutorbin.com/questions-and-answers/propylene-c3h6-is-a-valuable-feed-stock-which-is-commonly-produced-in-
0 Response to "To Mass Produce Propane C3h8 a Continuously Stirred Tank Reactor Cstr Is Used"
Post a Comment